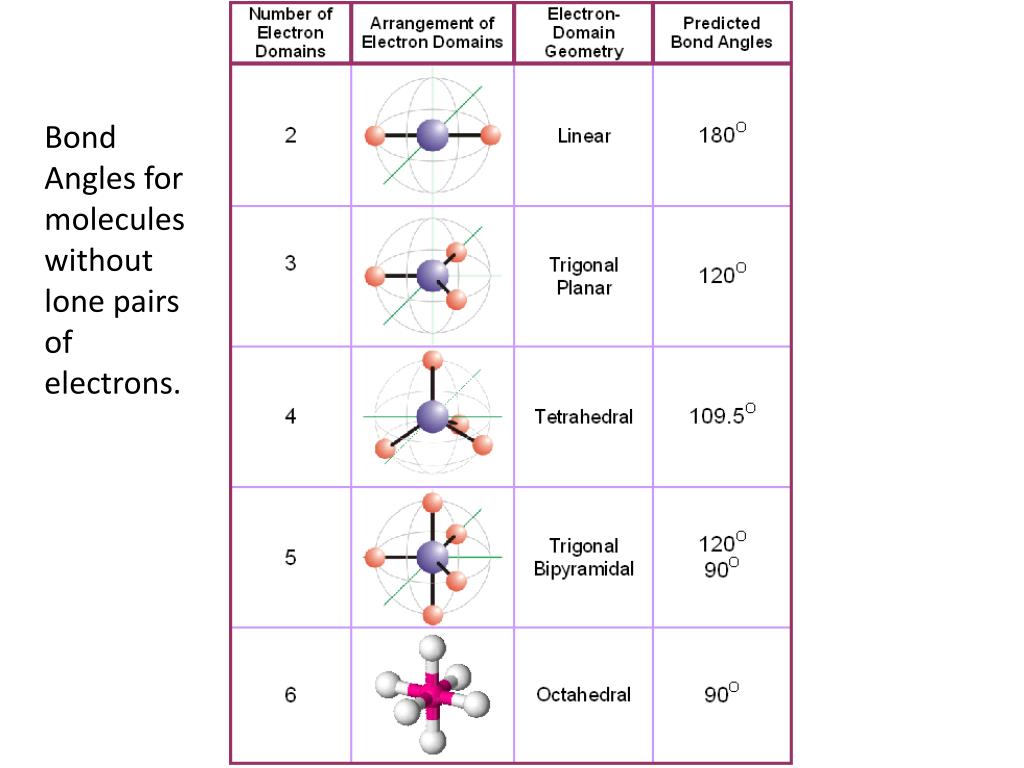
However, if there are lone pairs on a linear shape, I just memorize it is always 180 degrees. If a molecular geometry shape has lone pairs, the bond angle will have a less than sign since the lone pairs push down on the terminal atoms, decreasing the bond angle. For the 6th row, I memorize that all of them have 90 degree angles. For the 5th row, I memorize that the first two have 120 degree and 90 degree angles, but then a T shape has 90 degree angles (based on its shape). 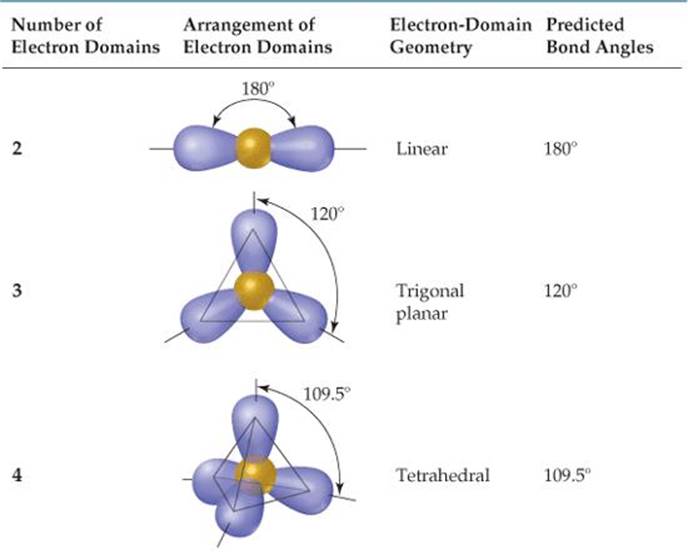
If there are three, then it is 120 degrees. This is the chart I use as well! For me, I memorize how many electron densities (number of terminal atoms + number of lone pairs). And as previously mentioned, linear is always 180 degrees, so a geometry with 6 electron domains and 4 lone pairs is 180 degrees. If you are having trouble with the last row, notice that the angles are 90 degrees, < 90 degrees, 90 degrees, and <90 degrees respectively. It is an oxyanion of iodine produced by the deprotonation of iodous acid (HIO 2 ). Iodite ion, also known as dioxidoiodate is represented by the chemical formula IO 2. The way I committed these to memory was by remembering that linear always equates to 180 degrees and that T-shape always equates to <90 degrees. Iodite (IO2-) lewis dot structure, molecular geometry or shape, electron geometry, bond angles, hybridization, polar or nonpolar. The discrepancies to this pattern lie toward the bottom of the table which contains molecules of 5 or 6 domains with 2 to 4 lone pairs. For a VSEPR model with four electron domains, you simply need to know that 2 lone pairs require an extra (<<) symbol, which makes sense because the more electrons there are, the greater distortion of the bond angle. Every bond angle value listed under 1 lone pair is always denoted as less than (<) the values listed in the basic geometries from which they were derived. As you can see below, the basic geometries (0 lone pairs) of each model all have solid values of 180 degrees, 109.5 degrees, etc. The way I memorized the bond angles was by finding a pattern within the VSEPR geometry chart.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
